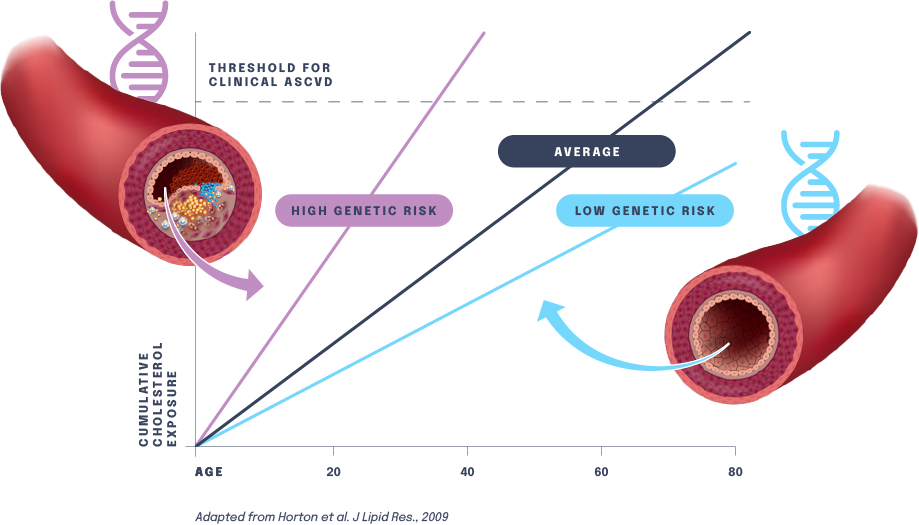
Human genetic studies have shown that certain individuals have gene variants that cause elevated blood levels of low-density lipoprotein cholesterol (LDL-C), leading to increased risk for ASCVD and ASCVD-related outcomes such as heart attack. Conversely, studies have found that certain gene variants are associated with natural disease resistance and reduce the risk for ASCVD. For instance, a subset of the population carries a naturally protective variant of a gene known as PCSK9. People born with this variant have extremely low LDL-C levels and are heart-healthy and resistant to heart attack.
Our gene editing medicines are designed to mimic the protective effects of naturally occurring resistance mutations. By potently and durably lowering cumulative LDL-C exposure throughout a patient’s lifetime, our gene editing medicines could fundamentally disrupt the chronic care model for treating patients with or at risk for ASCVD and relieve the significant burden placed on patients, providers and the healthcare system.